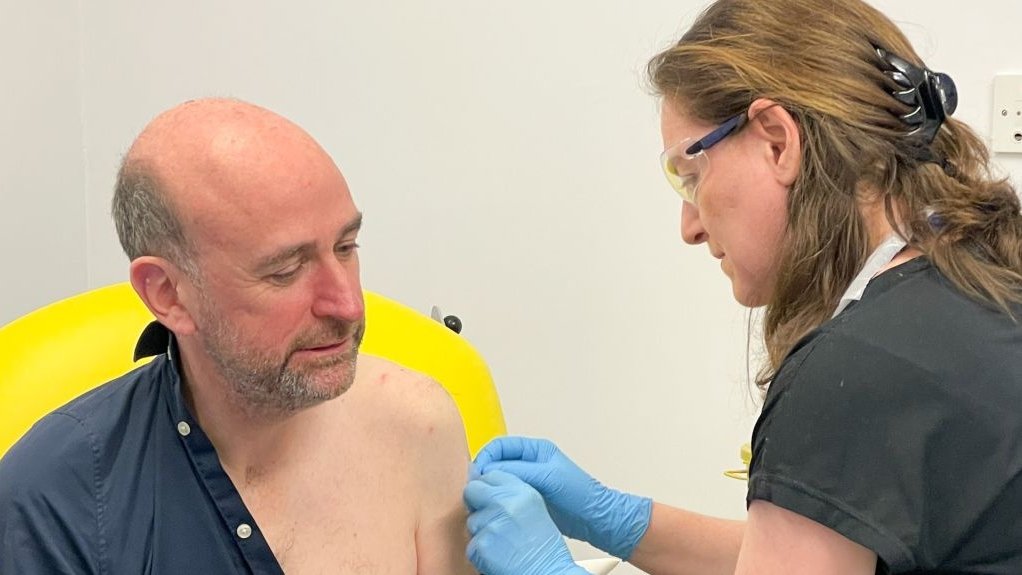
The Oxford Vaccine Group is embarking on an innovative venture as they undertake human trials for the ChAdOx1 NipahB vaccine, a potential solution to the devastating Nipah virus. This marks a significant advancement in medical research as the Nipah virus, which has a high mortality rate and is classified as a priority disease by the World Health Organization, currently lacks any approved vaccines or treatments.
Introducing the Nipah Virus
The Nipah virus is a zoonotic virus that is transmitted from animals to humans. Fruit bats serve as the primary natural host of this lethal disease. However, it can also be transmitted through contaminated food or direct human-to-human contact. Infections can range from asymptomatic cases to severe respiratory infections and fatal encephalitis.
ChAdOx1 NipahB Vaccine Trials: An Anchor of Hope
The ChAdOx1 NipahB vaccine trials involve 51 participants, all between the ages of 18 and 55. The trials are expected to span 18 months, overseen by the diligent Oxford Vaccine Group. Notably, this vaccine employs the ChAdOx1 platform, also used for the Oxford/AstraZeneca COVID-19 vaccine. Subsequent trials are planned to occur in a country affected by the Nipah virus.
Implications for Public Health
The development of a Nipah virus vaccine is not solely a scientific milestone but also holds significant implications for public health. The virus has a profound impact on several Asian countries, including India, where outbreaks have persisted for the past 25 years. Successful outcomes from these trials could establish a resilient defense against this lethal disease.