Eyestem Research Submits Pivotal IND Application for Novel Therapy Eyecyte RPE
Eyestem Research, an innovative deep science venture supported by DBT BIRAC, has recently submitted an Investigational New Drug (IND) application in India for their latest invention, Eyecyte RPE. This important development is facilitated by the Central Drugs Standards Control Organization (CDSCO) and heralds the possibility of first-in-human trials occurring soon.
The Company on a Mission with Groundbreaking Research
Steered by their dedicated CEO, Jogin Desai, Eyestem Research has been intensively involved in exhaustive research and crucial discussions with various regulatory bodies. Our source reports these important dialogues, designed to meet international standards for therapeutic approval, occurred across several territories, including India, Australia, Singapore, and the United States.
The Complex Path to Drug Approval
The journey towards gaining authorization for a new drug is replete with intricate processes. These involve the collection and presentation of an enormous volume of data alongside numerous documents. This cumbersome process aims to guarantee the safety and effectiveness of the novel therapies. Notably, regulatory processes vary globally with India emphasizing input controls and countries like Australia prioritizing output controls.
Eyestem’s Significant Achievement with IND Approval
As reported by our source, Eyestem’s recent IND approval is a noteworthy milestone. This achievement signifies years of diligent research, and it represents the onset of impending clinical trials. Despite variations in country-specific conditions, Eyestem’s journey through the complex regulation landscape underlines the ongoing worldwide efforts towards harmonization.
Aiming for Economic Accessibility and Broadened Scope
In addition to their scientific breakthroughs, Eyestem Research is also committed to making their unique therapies economically accessible. They are aiming to set a pricing standard that undercuts the cost of current cell and gene therapies significantly. Furthermore, the company is widening its research horizons and exploring treatments for other afflictions such as Parkinson’s disease and retinitis pigmentosa.
Conclusion: A Milestone for Eyestem and Hope for Patients
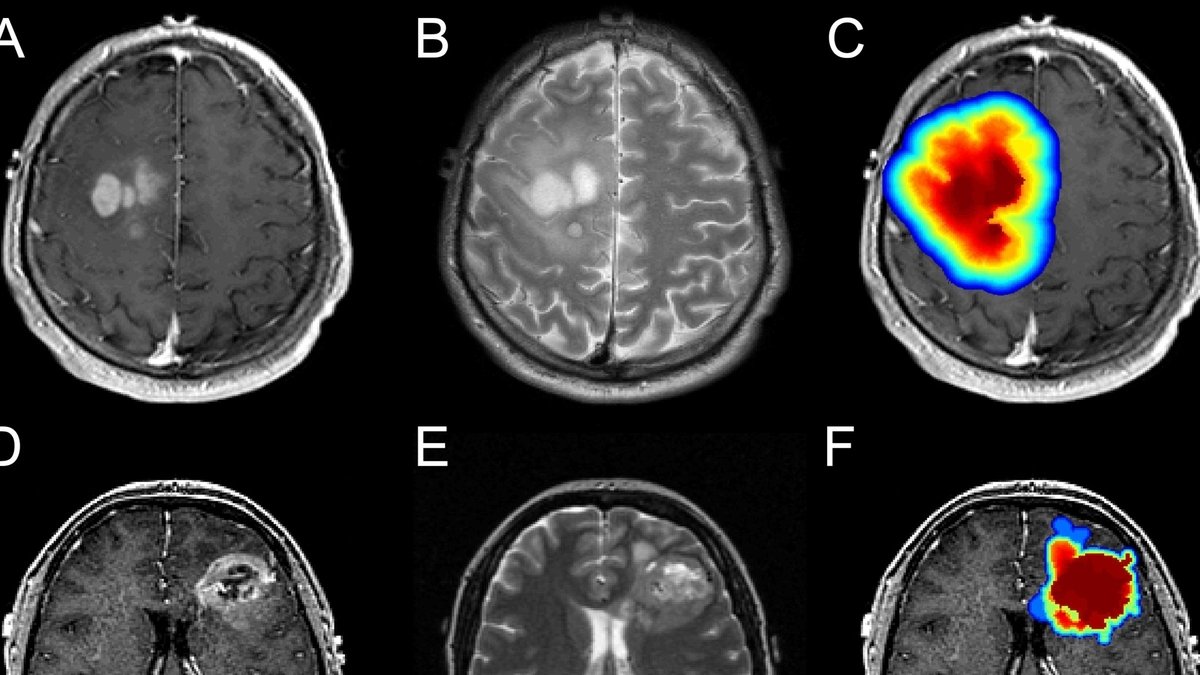
With the submission of their IND application for Eyecyte RPE, Eyestem Research marks a significant milestone in their journey. This development not only represents the culmination of years of dedicated research but also offers hope for patients of geographic atrophy secondary to dry AMD. While the path to global regulatory approval may vary, the company’s commitment to affordable treatment options underscores their mission to bring meaningful change to patients’ lives.